
0 is he permittivity constant: 0 =8.85 x 10 -12 C 2/(N. R is the distance between particle 1 and particle 2 (see Figure 22.1) Q 1 and q 2 are the charges of particle 1 and particle 2,respectively Electric charges of the building blocks of atoms The direction of theforce is along the line joining the particles. " The magnitude of the electric force that a particle exerts on anotherparticle is directly proportional to the product of their charges and inverselyproportional to the square of the distance between them. The precise magnitude of the electric force that a charged particle exerts onanother is given by Coulomb's law: Since atoms are neutral, the number of electronsmust be equal to the number of protons. Detailed measurements have shown thatthe magnitude of the charge of the proton is exactly equal to the magnitude ofthe charge of the electron. The electric charge of electrons, protonsand neutrons are listed in Table 22.2. Per definition, the electric charge on a glassrod rubbed with silk is positive. The electric charge can benegative, zero, or positive. The unit ofelectric charge q is the Coulomb (C). Instead,it depends on a new quantity: the electric charge. This impliesthat the electric force does not depend on the mass of the particle. 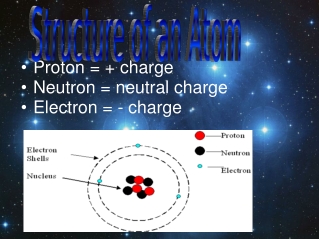
The attractive force between the electronsand the nucleus is called the electric force.Įxperiments have shown that the electric force between two objects isproportional to the inverse square of the distance between the two objects.The electric force between two electrons is the same as the electric forcebetween two protons when they are placed as the same distance. Measurements of the velocity of theorbital electrons in an atom have shown that the attractive force between theelectrons and the nucleus is significantly stronger than the gravitationalforce between these two objects. In P121 it was shown that anobject can only carry out circular motion if a radial force (directed towardsthe center of the circle) is present. The electrons are contained in a roughly spherical regionwith a diameter of about 2 x 10 -10 m. The diameter of the nucleus is between 10 -15 and10 -14 m. Mostof the mass of the atom is due to the mass of the nucleus. Themasses of the electrons, protons and neutrons are listed in Table 22.1. Each atom consists of a nucleus,consisting of protons and neutrons, surrounded by a number of electrons. 
Charge Quantization and Charge Conservation using Avogadro’s number, using shot noise method etc.ELECTRIC FORCE AND ELECTRIC CHARGE CHAPTER 22 Apart from this, there are many other methods to find out the fundamental value of electric charge, e.g.Repeating the same for different drops, it can be seen that, every time the value of the charge is integral multiple of the elementary charge. Now we know that electric force on a charge is the product of the charge and the field in which it resides, since we know the field, by dividing the force by the field we can accurately determine the value of the charge in the oil drop. Electric force on it can also be deduced. Now, the forces on the drop due to gravity and viscosity could be deducted by measuring the speed of the drop and the size of the drop. The forces that would be working on the drop are the gravitational force on it due to its weight, the force due to viscosity as it is falling through air and the electrostatic force on the charges in it due to the electric field. Millikan’s oil drop experiment- In the experiment a small drop of oil was dropped from a height in an electric field.The theory behind the experiment was quite simple and yet brilliant. Millikan, through his famous oil drop experiment first measured the fundamental unit of electric charge.This is also not a violation of the fundamental unit of charge because quasiparticles are not actually elementary particles. 
In the quantum Hall effect theory of Robert Laughlin, it is shown that the quasiparticles have fractional charges to that of the elementary charge. On the other hand, quasiparticles are not actually particles but an emergent entity that behaves as particles. Thus, we can justify that our independent fundamental unit of charge is e. But quarks cannot exist in an isolated form. Quarks have charge in multiples of 1 3 e \frace 3 1 e. But there are two cases where it can be thought of as exceptions (but actually they are not). Elementary charge is basically indivisible. So charges can exist in the form of 0e or 1e or 2e. This principle of any amount of charge being an integral multiple of elementary charge is called Charge quantization. That is the reason behind the terminology, elementary charge. It is called the fundamental unit of charge because charge contained in any object is found to be integral multiple of it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
